iCellar™

Proprietary supplements and/or engineered cells for promoting robust cell expansion capability while maintaining high cell quality

One of the main challenges of cell therapy development is generating sufficient number of high quality (functional) therapeutic cells in timely fashion. In autologous CAR T cell therapy, hundreds of millions (1x10⁸) to billions (1x10⁹) of cells are often required for each infusion to patients. Prolonged culture is therefore necessary to achieve sufficient cell production. However, cells tend to undergo senescence, exhaustion, and differentiation in prolonged culture leading to reduction of anti-tumor efficacy. Hence, a robust cell culture system for manufacturing clinical-scaled and functional genetically engineered cells is needed.
GenomeFrontier overcomes this challenge by introducing iCellar™ , a platform composed of proprietary agents and/or gene-modified cells that are designed to enhance cell expansion capacity while maintaining cell quality.
Upon antigen stimulation, naïve (TN) cells first differentiate into TSCM cells and may further differentiate into short-lived effector TEFF, the most differentiated and exhausted T cell subset. It has been clinically demonstrated that high amounts of CAR⁺ TSCM cells lead to better clinical outcomes due to the high metabolic fitness, stemness, and long-term persistence of this cell subset as illustrated in Figure 1.
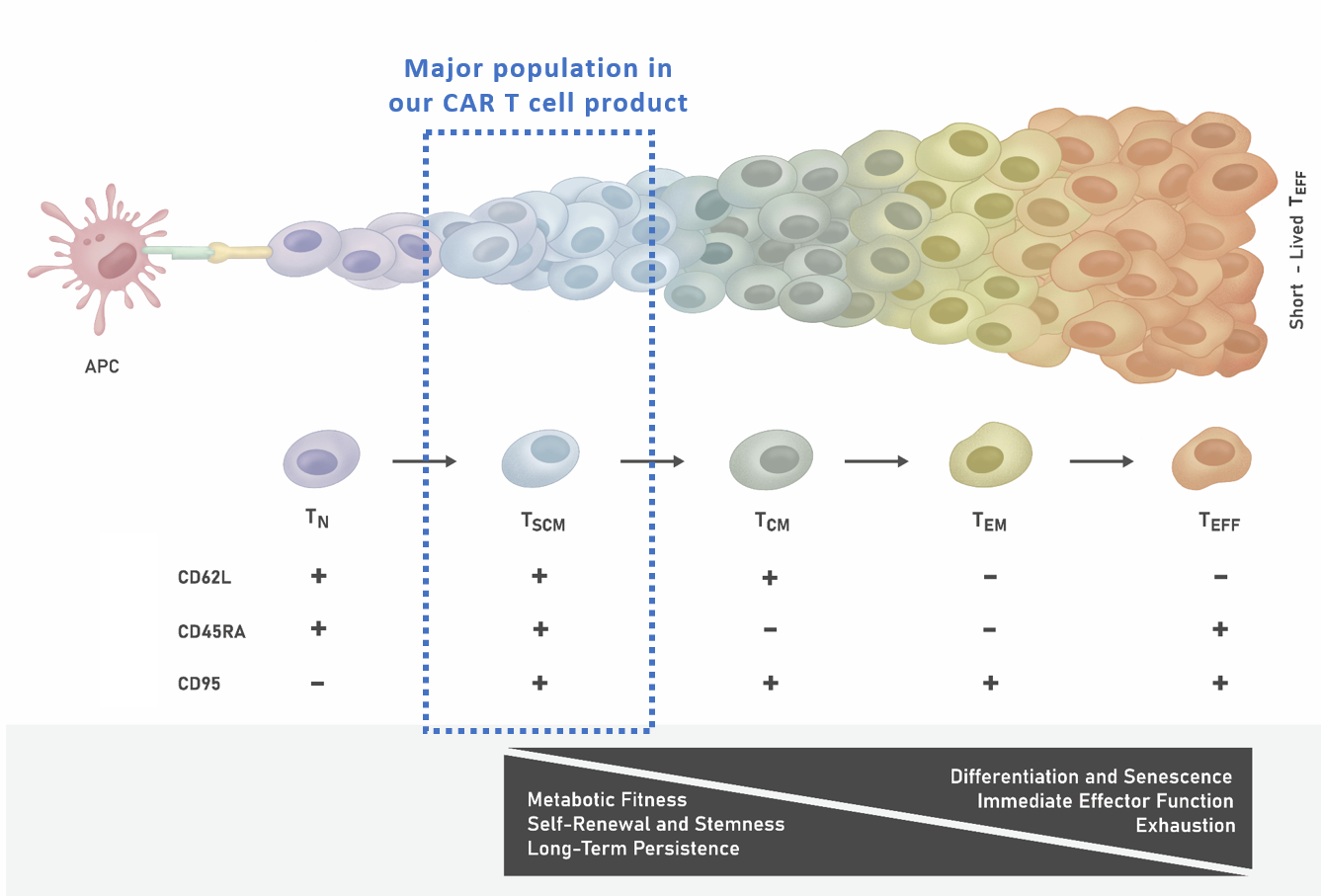
Figure 1. T Cell Differentiation Stages Upon Antigen Stimulation
Following the activation of TN cells by tumor associated antigens (presented on antigen presenting cells, or APC), long-lived TSCM and TCM are produced. These self-renewing T cells in turn give rise to proliferating populations of shorter-lived TEM and also TEFF cells. TSCM cells are thought to be the most therapeutically efficacious T cell population.
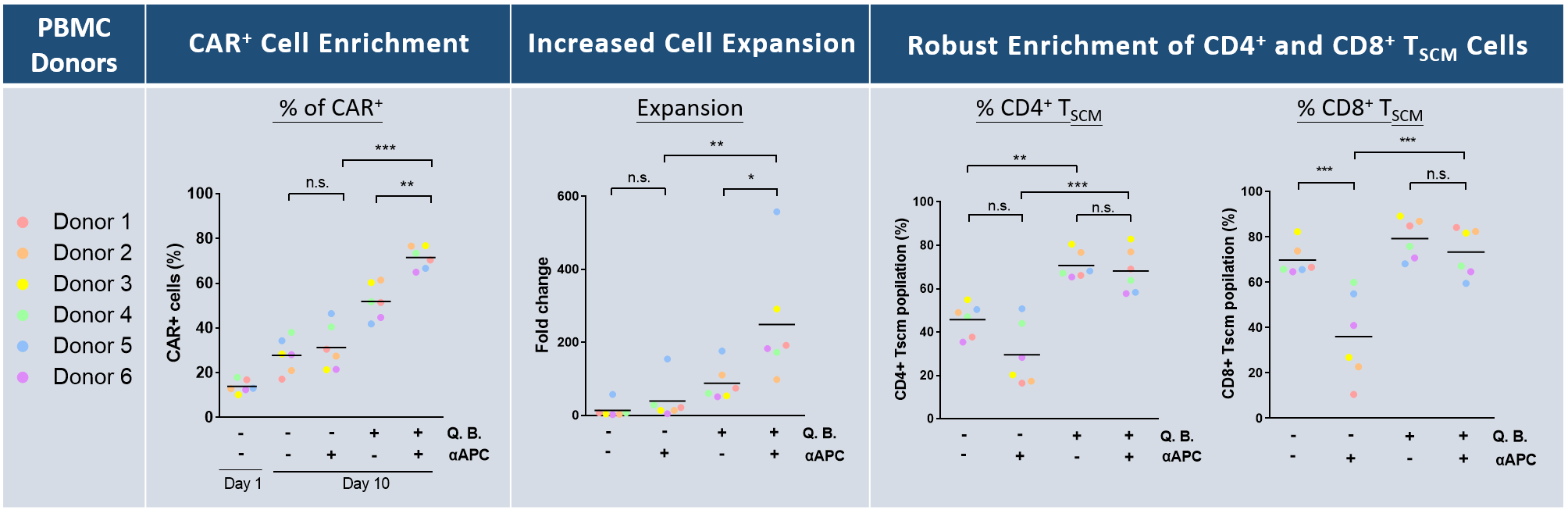
For timely generation of clinical scaled CAR⁺ TSCM cells , we have successfully developed iCellar™ system composed of two components: (1) Artificial antigen presenting cells (aAPCs) that enrich and expand CAR⁺ T cells; and (2)Quantum Booster™ which robustly expand TSCM cells. Combining these two agents (1) markedly enriched CAR⁺ T cell subpopulation; (2) with up to 90% of CD4⁺/CAR⁺ and CD8⁺/CAR⁺ populations being TSCM cells; and (3) increased expansion of CAR-T cells while preserving quality (Figure 2). Within ten days of culture, iCellar™ enables production of high quality CAR⁺ TSCM cell population in clinical scale (~1x10⁹).