Defining Unmet Needs in Gene Therapy
Gene therapy is a technique that prevents, treats, or cures disease via gene modification of patient cells. One type of gene therapy is cell-modified gene therapy, which involves removing cells from the patient, genetically modifying the cells using a gene delivery/modification vehicle, and reinfusing the reengineered cells back to the patient.
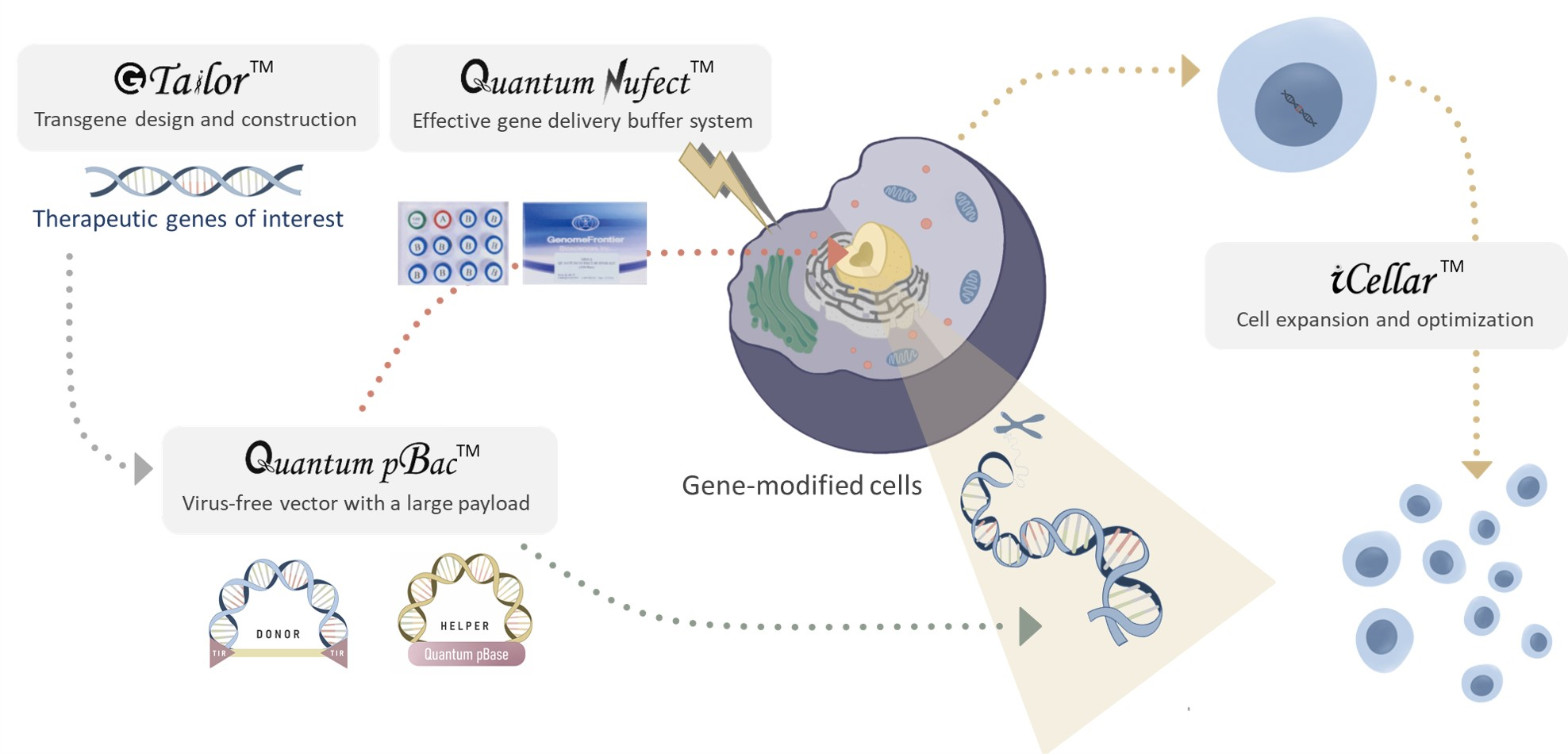
Viral vector is currently the most common vehicle for gene therapy due to its high gene delivery efficiency. However, using viral vectors has several disadvantages, including: (1) virus-associated safety risks, (2) high manufacturing cost, and (3) limited gene payload capacity, severely restricting the repertoire of genes that can be encoded.
A virus-free vector is a safer alternative to viral vectors due to lower immunogenicity and lower risk of genome toxicity. Moreover, virus-free vector has the capacity to carry larger fragments of genetic material compared to conventional viral vectors. The manufacturing costs of virus-free vectors are also markedly lower than those required for viral vector production.
Despite the advantages of virus-free vectors, it remains a challenge to efficiently modify, expand, and deliver persistent therapeutic cells with stable gene expression. Electroporation is the most efficient method for delivering virus-free vectors into therapeutic cells, but results in fragile cells with markedly reduced viability and expansion capacity.